Black Beans vs Pinto Beans – Which is Healthier?
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Our Verdict
When comparing black beans to pinto beans, we picked the pinto beans.
Why?
Both of these beans have won all their previous comparisons, so it’s no surprise that this one was very close. Despite their different appearance, taste, and texture, their nutritional profiles are almost identical:
In terms of macros, pinto beans have a tiny bit more protein, carbs, and fiber. So, a nominal win for pinto beans, but again, the difference is very slight.
When it comes to vitamins, black beans have more of vitamins A, B1, B3, and B5, while pinto beans have more of vitamins B2, B6, B9, C, E, K and choline. Superficially, again this is nominally a win for pinto beans, but in most cases the differences are so slight as to be potentially the product of decimal place rounding.
In the category of minerals, black beans have more calcium, copper, iron, and phosphorus, while pinto beans have more magnesium, manganese, selenium, and zinc. That’s a 4:4 tie, but the only one with a meaningful margin of difference is selenium (of which pinto beans have 4x more), so we’re calling this one a very modest win for pinto beans.
All in all, adding these up makes for a “if we really are pressed to choose” win for pinto beans, but honestly, enjoy either in accordance with your preference (this writer prefers black beans!), or better yet, both.
Want to learn more?
You might like to read:
What’s Your Plant Diversity Score?
Take care!
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Recommended
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails:
-
Struggle To Deep Squat? It’s Probably This One Fixable Thing Holding You Back
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Deep squat, Asian squat, Slav squat, resting squat… Whatever we want to call it, many in the West struggle with it.
Nevertheless, this struggle is entirely circumstantial, very fixable, and definitely not some kind of immutable law of the universe:
A strong foundation
A proper deep squat relies on four main factors:
- Ankle dorsiflexion allows the toes to pull toward the shin.
- Hip flexion and external rotation bring the thighs close to the torso while slightly rotating outward.
- Knee flexion ensures the thighs and calves make contact.
- Maintaining the center of mass over the midfoot is essential for balance.
Correspondingly, the reason for struggling can be a case of…
- Limited ankle mobility, which prevents the knees from moving forward, shifting weight backward.
- Tight glutes and weak hip flexors making it hard to bring the torso close to the thighs, often causing people to fall backward.
- Quad tightness can also restrict depth if the thighs cannot meet the calves.
- Proportionally longer femurs than average can cause extra difficulty as the pelvis shifts further back, requiring more knee travel for balance.
However, we said “one thing”, not “four things”, so what’s the deal?
For most people, we are told in this video, ankle mobility is the biggest limiting factor in achieving a deep squat. Thus, she recommends working on that, and (at the end of this video) links to another video with specifically ankle exercises.
For all of this plus visual demonstrations, enjoy:
Click Here If The Embedded Video Doesn’t Load Automatically!
Want to learn more?
You might also like:
The Secret To Better Squats: Foot, Knee, & Ankle Mobility
Take care!
Share This Post
-
Dogs Paired With Providers at Hospitals Help Ease Staff and Patient Stress
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
DENVER — Outside HCA HealthONE Rose medical center, the snow is flying. Inside, on the third floor, there’s a flurry of activity within the labor and delivery unit.
“There’s a lot of action up here. It can be very stressful at times,” said Kristina Fraser, an OB-GYN in blue scrubs.
Nurses wheel a very pregnant mom past.
“We’re going to bring a baby into this world safely,” Fraser said, “and off we go.”
She said she feels ready in part due to a calming moment she had just a few minutes earlier with some canine colleagues.
A pair of dogs, tails wagging, had come by a nearby nursing station, causing about a dozen medical professionals to melt into a collective puddle of affection. A yellow Lab named Peppi showered Fraser in nuzzles and kisses. “I don’t know if a human baby smells as good as that puppy breath!” Fraser had said as her colleagues laughed.
The dogs aren’t visitors. They work here, too, specifically for the benefit of the staff. “I feel like that dog just walks on and everybody takes a big deep breath and gets down on the ground and has a few moments of just decompressing,” Fraser said. “It’s great. It’s amazing.”
Hospital staffers who work with the dogs say there is virtually no bite risk with the carefully trained Labradors, the preferred breed for this work.
The dogs are kept away from allergic patients and washed regularly to prevent germs from spreading, and people must wash their hands before and after petting them.
Doctors and nurses are facing a growing mental health crisis driven by their experiences at work. They and other health care colleagues face high rates of depression, anxiety, stress, suicidal ideation, and burnout. Nearly half of health workers reported often feeling burned out in 2022, an increase from 2018, according to the Centers for Disease Control and Prevention. And the percentage of health care workers who reported harassment at work more than doubled over that four-year period. Advocates for the presence of dogs in hospitals see the animals as one thing that can help.
That includes Peppi’s handler, Susan Ryan, an emergency medicine physician at Rose.
Ryan said years working as an emergency room doctor left her with symptoms of PTSD. “I just was messed up and I knew it,” said Ryan, who isolated more at home and didn’t want to engage with friends. “I shoved it all in. I think we all do.”
She said doctors and other providers can be good at hiding their struggles, because they have to compartmentalize. “How else can I go from a patient who had a cardiac arrest, deal with the family members telling them that, and go to a room where another person is mad that they’ve had to wait 45 minutes for their ear pain? And I have to flip that switch.”
To cope with her symptoms of post-traumatic stress disorder, Ryan started doing therapy with horses. But she couldn’t have a horse in her backyard, so she got a Labrador.
Ryan received training from a national service dog group called Canine Companions, becoming the first doctor trained by the group to have a facility dog in an emergency room. Canine Companions has graduated more than 8,000 service dogs.
The Rose medical center gave Ryan approval to bring a dog to work during her ER shifts. Ryan’s colleagues said they are delighted that a dog is part of their work life.
“When I have a bad day at work and I come to Rose and Peppi is here, my day’s going to be made better,” EMT Jasmine Richardson said. “And if I have a patient who’s having a tough day, Peppi just knows how to light up the room.”
Nursing supervisor Eric Vaillancourt agreed, calling Peppi “joyful.”
Ryan had another dog, Wynn, working with her during the height of the pandemic. She said she thinks Wynn made a huge difference. “It saved people,” she said. “We had new nurses that had never seen death before, and now they’re seeing a covid death. And we were worried sick we were dying.”
She said her hospital system has lost a couple of physicians to suicide in the past two years, which HCA confirmed to KFF Health News and NPR. Ryan hopes the canine connection can help with trauma. “Anything that brings you back to the present time helps ground you again. A dog can be that calming influence,” she said. “You can get down on the ground, pet them, and you just get calm.”
Ryan said research has shown the advantages. For example, one review of dozens of original studies on human-animal interactions found benefits for a variety of conditions including behavioral and mood issues and physical symptoms of stress.
Rose’s president and CEO, Casey Guber, became such a believer in the canine connection he got his own trained dog to bring to the hospital, a black Lab-retriever mix named Ralphie.
She wears a badge: Chief Dog Officer.
Guber said she’s a big morale booster. “Phenomenal,” he said. “It is not uncommon to see a surgeon coming down to our administration office and rolling on the ground with Ralphie, or one of our nurses taking Ralphie out for a walk in the park.”
This article is from a partnership that includes CPR News, NPR, and KFF Health News.
KFF Health News is a national newsroom that produces in-depth journalism about health issues and is one of the core operating programs at KFF—an independent source of health policy research, polling, and journalism. Learn more about KFF.
Subscribe to KFF Health News’ free Morning Briefing.
This article first appeared on KFF Health News and is republished here under a Creative Commons license.
Share This Post
-
Ovarian cancer is hard to detect. Focusing on these 4 symptoms can help with diagnosis
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Ovarian cancers are often found when they are already advanced and hard to treat.
Researchers have long believed this was because women first experienced symptoms when ovarian cancer was already well-established. Symptoms can also be hard to identify as they’re vague and similar to other conditions.
But a new study shows promising signs ovarian cancer can be detected in its early stages. The study targeted women with four specific symptoms – bloating, abdominal pain, needing to pee frequently, and feeling full quickly – and put them on a fast track to see a specialist.
As a result, even the most aggressive forms of ovarian cancer could be detected in their early stages.
So what did the study find? And what could it mean for detecting – and treating – ovarian cancer more quickly?
Ground Picture/Shutterstock Why is ovarian cancer hard to detect early?
Ovarian cancer cannot be detected via cervical cancer screening (which used to be called a pap smear) and pelvic exams aren’t useful as a screening test.
Current Australian guidelines recommend women get tested for ovarian cancer if they have symptoms for more than a month. But many of the symptoms – such as tiredness, constipation and changes in menstruation – are vague and overlap with other common illnesses.
This makes early detection a challenge. But it is crucial – a woman’s chances of surviving ovarian cancer are associated with how advanced the cancer is when she is diagnosed.
If the cancer is still confined to the original site with no spread, the five-year survival rate is 92%. But over half of women diagnosed with ovarian cancer first present when the cancer has already metastatised, meaning it has spread to other parts of the body.
If the cancer has spread to nearby lymph nodes, the survival rate is reduced to 72%. If the cancer has already metastasised and spread to distant sites at the time of diagnosis, the rate is only 31%.
There are mixed findings on whether detecting ovarian cancer earlier leads to better survival rates. For example, a trial in the UK that screened more than 200,000 women failed to reduce deaths.
That study screened the general public, rather than relying on self-reported symptoms. The new study suggests asking women to look for specific symptoms can lead to earlier diagnosis, meaning treatment can start more quickly.
What did the new study look at?
Between June 2015 and July 2022, the researchers recruited 2,596 women aged between 16 and 90 from 24 hospitals across the UK.
They were asked to monitor for these four symptoms:
- persistent abdominal distension (women often refer to this as bloating)
- feeling full shortly after starting to eat and/or loss of appetite
- pelvic or abdominal pain (which can feel like indigestion)
- needing to urinate urgently or more often.
Women who reported at least one of four symptoms persistently or frequently were put on a fast-track pathway. That means they were sent to see a gynaecologist within two weeks. The fast track pathway has been used in the UK since 2011, but is not specifically part of Australia’s guidelines.
Some 1,741 participants were put on this fast track. First, they did a blood test that measured the cancer antigen 125 (CA125). If a woman’s CA125 level was abnormal, she was sent to do a internal vaginal ultrasound.
What did they find?
The study indicates this process is better at detecting ovarian cancer than general screening of people who don’t have symptoms. Some 12% of women on the fast-track pathway were diagnosed with some kind of ovarian cancer.
A total of 6.8% of fast-tracked patients were diagnosed with high-grade serous ovarian cancer. It is the most aggressive form of cancer and responsible for 90% of ovarian cancer deaths.
Out of those women with the most aggressive form, one in four were diagnosed when the cancer was still in its early stages. That is important because it allowed treatment of the most lethal cancer before it had spread significantly through the body.
There were some promising signs in treating those with this aggressive form. The majority (95%) had surgery and three quarters (77%) had chemotherapy. Complete cytoreduction – meaning all of the cancer appears to have been removed – was achieved in six women out of ten (61%).
It’s a promising sign that there may be ways to “catch” and target ovarian cancer before it is well-established in the body.
What does this mean for detection?
The study’s findings suggest this method of early testing and referral for the symptoms leads to earlier detection of ovarian cancer. This may also improve outcomes, although the study did not track survival rates.
It also points to the importance of public awareness about symptoms.
Clinicians should be able to recognise all of the ways ovarian cancer can present, including vague symptoms like general fatigue.
But empowering members of the general public to recognise a narrower set of four symptoms can help trigger testing, detection and treatment of ovarian cancer earlier than we thought.
This could also save GPs advising every woman who has general tiredness or constipation to undergo an ovarian cancer test, making testing and treatment more targeted and efficient.
Many women remain unaware of the symptoms of ovarian cancer. This study shows recognising them may help early detection and treatment.
Jenny Doust, Clinical Professorial Research Fellow, Australian Women and Girls’ Health Research Centre, The University of Queensland
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Share This Post
Related Posts
-
Semaglutide’s Surprisingly Unexamined Effects
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Semaglutide’s Surprisingly Big Research Gap
GLP-1 receptor agonists like Ozempic, Wegovy, and other semaglutide drugs. are fast becoming a health industry standard go-to tool in the weight loss toolbox. When it comes to recommending that patients lose weight, “Have you considered Ozempic?” is the common refrain.
Sometimes, this may be a mere case of kicking the can down the road with regard to some other treatment that it can be argued (sometimes even truthfully) would go better after some weight loss:
How weight bias in health care can harm patients with obesity: Research
…which we also covered in fewer words in the second-to-last item here:
But GLP-1 agonists work, right?
Yes, albeit there’s a litany of caveats, top of which are usually:
- there are often adverse gastrointestinal side effects
- if you stop taking them, weight regain generally ensues promptly
For more details on these and more, see:
…but now there’s another thing that’s come to light:
The dark side of semaglutide’s weight loss
In academia, “dark” is often used to describe “stuff we don’t have much (or in some cases, any) direct empirical evidence of, but for reasons of surrounding things, we know it’s there”.
Well-known examples include “dark matter” in physics and the Dark Ages in (European) history.
In the case of semaglutide and weight loss, a review by a team of researchers (Drs. Sandra Christenen, Katie Robinson, Sara Thomas, and Dominique Williams) has discovered how little research has been done into a certain aspect of GLP-1 agonist’s weight loss effects, namely…
Dietary changes!
There’s been a lot of popular talk about “people taking semaglutide eat less”, but it’s mostly anecdotal and/or presumed based on parts of the mechanism of action (increasing insulin production, reducing glucagon secretions, modulating dietary cravings).
Where studies have looked at dietary changes, it’s almost exclusively been a matter of looking at caloric intake (which has been found to be a 16–39% reduction), and observations-in-passing that patients reported reduction in cravings for fatty and sweet foods.
This reduction in caloric intake, by the way, is not significantly different to the reduction brought about by counselling alone (head-to-head studies have been done; these are also discussed in the research review).
However! It gets worse. Very few studies of good quality have been done, even fewer (two studies) actually had a registered dietitian nutritionist on the team, and only one of them used the “gold standard” of nutritional research, the 24-hour dietary recall test. Which, in case you’re curious, you can read about what that is here:
Dietary Assessment Methods: What Is A 24-Hour Recall?
Of the four studies that actually looked at the macros (unlike most studies), they found that on average, protein intake decreased by 17.1%. Which is a big deal!
It’s an especially big deal, because while protein’s obviously important for everyone, it’s especially important for anyone trying to lose weight, because muscle mass is a major factor in metabolic base rate—which in turn is much important for fat loss/maintenance than exercise, when it comes to how many calories we burn by simply existing.
A reasonable hypothesis, therefore, is that one of the numerous reasons people who quit GLP-1 agonists immediately put fat back on, is because they probably lost muscle mass in amongst their weight loss, meaning that their metabolic base rate will have decreased, meaning that they end up more disposed to put on fat than before.
And, that’s just a hypothesis and it’s a hypothesis based on very few studies, so it’s not something to necessarily take as any kind of definitive proof of anything, but it is to say—as the researchers of this review do loudly say—more research needs to be done into this, because this has been a major gap in research so far!
Any other bad news?
While we’re talking research gaps, guess how many studies looked into micronutrient intake changes in people taking GLP-1 agonists?
If you guessed zero, you guessed correctly.
You can find the paper itself here:
What’s the main take-away here?
On a broad, scoping level: we need more research!
On a “what this means for individuals who want to lose weight” level: maybe we should be more wary of this still relatively new (less than 10 years old) “wonder drug”. And for most of those 10 years it’s only been for diabetics, with weight loss use really being in just the past few years (2021 onwards).
In other words: not necessarily any need to panic, but caution is probably not a bad idea, and natural weight loss methods remain very reasonable options for most people.
See also: How To Lose Weight (Healthily!)
Take care!
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails:
-
The Brain As A Work-In-Progress
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
And The Brain Goes Marching On!
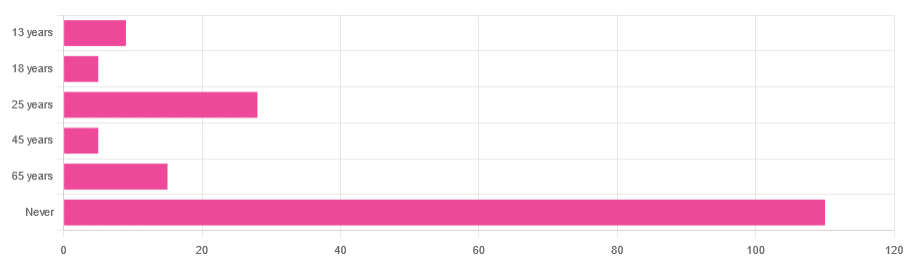
In Tuesday’s newsletter, we asked you “when does the human brain stop developing?” and got the above-depicted, below-described, set of responses:
- About 64% of people said “Never”
- About 16% of people said “25 years”
- About 9% of people said “65 years”
- About 5% of people said “13 years”
- About 3% of people said “18 years”
- About 3% of people said “45 years”
Some thoughts, before we get into the science:
An alternative wording for the original question was “when does the human brain finish developing”; the meaning is the same but the feeling is slightly different:
- “When does the human brain stop developing?” focuses attention on the idea of cessation, and will skew responses to later ages
- When does the human brain finish developing?” focuses on attention on a kind of “is it done yet?” and will skew responses to earlier ages
Ultimately, since we had to chose one word or another, we picked the shortest one, but it would have been interesting if we could have done an A/B test, and asked half one way, and half the other way!
Why we picked those ages
We picked those ages as poll options for reasons people might be drawn to them:
- 13 years: in English-speaking cultures, an important milestone of entering adolescence (note that the concept of a “teenager” is not precisely universal as most languages do not have “-teen” numbers in the same way; the concept of “adolescent” may thus be tied to other milestones)
- 18 years: age of legal majority in N. America and many other places
- 25 years: age popularly believed to be when the brain is finished developing, due to a study that we’ll talk about shortly (we guess that’s why there’s a spike in our results for this, too!)
- 45 years: age where many midlife hormonal changes occur, and many professionals are considered to have peaked in competence and start looking towards retirement
- 65 years: age considered “senior” in much of N. America and many other places, as well as the cut-off and/or starting point for a lot of medical research
Notice, therefore, how a lot of things are coming from places they really shouldn’t. For example, because there are many studies saying “n% of people over 65 get Alzheimer’s” or “n% of people over 65 get age-related cognitive decline”, etc, 65 becomes the age where we start expecting this—because of an arbitrary human choice of where to draw the cut-off for the study enrollment!
Similarly, we may look at common ages of legal majority, or retirement pensions, and assume “well it must be for a good reason”, and dear reader, those reasons are more often economically motivated than they are biologically reasoned.
So, what does the science say?
Our brains are never finished developing: True or False?
True! If we define “finished developing” as “we cease doing neurogenesis and neuroplasticity is no longer in effect”.
Glossary:
- Neurogenesis: the process of creating new brain cells
- Neuroplasticity: the process of the brain adapting to changes by essentially rebuilding itself to suit our perceived current needs
We say “perceived” because sometimes neuroplasticity can do very unhelpful things to us (e.g: psychological trauma, or even just bad habits), but on a biological level, it is always doing its best to serve our overall success as an organism.
For a long time it was thought that we don’t do neurogenesis at all as adults, but this was found to be untrue:
How To Grow New Brain Cells (At Any Age)
Summary of conclusions of the above: we’re all growing new brain cells at every age, even if we be in our 80s and with Alzheimer’s disease, but there are things we can do to enhance our neurogenic potential along the way.
Neuroplasticity will always be somewhat enhanced by neurogenesis (after all, new neurons get given jobs to do), and we reviewed a great book about the marvels of neuroplasticity including in older age:
Our brains are still developing up to the age of 25: True or False?
True! And then it keeps on developing after that, too. Now this is abundantly obvious considering what we just talked about, but see what a difference the phrasing makes? Now it makes it sound like it stops at 25, which this statement doesn’t claim at all—it only speaks for the time up to that age.
A lot of the popular press about “the brain isn’t fully mature until the age of 25” stems from a 2006 study that found:
❝For instance, frontal gray matter volume peaks at about age 11.0 years in girls and 12.1 years in boys, whereas temporal gray matter volume peaks at about age at 16.7 years in girls and 16.2 years in boys. The dorsal lateral prefrontal cortex, important for controlling impulses, is among the latest brain regions to mature without reaching adult dimensions until the early 20s.❞
Source: Structural Magnetic Resonance Imaging of the Adolescent Brain
There are several things to note here:
- The above statement is talking about the physical size of the brain growing
- Nowhere does he say “and stops developing at 25”
However… The study only looked at brains up to the age of 25. After that, they stopped looking, because the study was about “the adolescent brain” so there has to be a cut-off somewhere, and that was the cut-off they chose.
This is the equivalent of saying “it didn’t stop raining until four o’clock” when the reality is that four o’clock is simply when you gave up on checking.
The study didn’t misrepresent this, by the way, but the popular press did!
Another 2012 study looked at various metrics of brain development, and found:
- Synapse overproduction into the teens
- Cortex pruning into the late 20s
- Prefrontal pruning into middle age at least (they stopped looking)
- Myelination beyond middle age (they stopped looking)
Source: Experience and the developing prefrontal cortex ← check out figure 1, and make sure you’re looking at the human data not the rat data
So how’s the most recent research looking?
Here’s a 2022 study that looked at 123,984 brain scans spanning the age range from mid-gestation to 100 postnatal years, and as you can see from its own figure 1… Most (if not all) brain-things keep growing for life, even though most slow down at some point, they don’t stop:
Brain charts for the human lifespan ← check out figure 1; don’t get too excited about the ventricular volume column as that is basically “brain that isn’t being a brain”. Do get excited about the rest, though!
Want to know how not to get caught out by science being misrepresented by the popular press? Check out:
How Science News Outlets Can Lie To You (Yes, Even If They Cite Studies!)
Take care!
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails:
-
No, taking drugs like Ozempic isn’t ‘cheating’ at weight loss or the ‘easy way out’
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Hundreds of thousands of people worldwide are taking drugs like Ozempic to lose weight. But what do we actually know about them? This month, The Conversation’s experts explore their rise, impact and potential consequences.
Obesity medication that is effective has been a long time coming. Enter semaglutide (sold as Ozempic and Wegovy), which is helping people improve weight-related health, including lowering the risk of a having a heart attack or stroke, while also silencing “food noise”.
As demand for semaglutide increases, so are claims that taking it is “cheating” at weight loss or the “easy way out”.
We don’t tell people who need statin medication to treat high cholesterol or drugs to manage high blood pressure they’re cheating or taking the easy way out.
Nor should we shame people taking semaglutide. It’s a drug used to treat diabetes and obesity which needs to be taken long term and comes with risks and side effects, as well as benefits. When prescribed for obesity, it’s given alongside advice about diet and exercise.
How does it work?
Semaglutide is a glucagon-like peptide-1 receptor agonist (GLP-1RA). This means it makes your body’s own glucagon-like peptide-1 hormone, called GLP-1 for short, work better.
GLP-1 gets secreted by cells in your gut when it detects increased nutrient levels after eating. This stimulates insulin production, which lowers blood sugars.
GLP-1 also slows gastric emptying, which makes you feel full, and reduces hunger and feelings of reward after eating.
GLP-1 receptor agonist (GLP-1RA) medications like Ozempic help the body’s own GLP-1 work better by mimicking and extending its action.
Some studies have found less GLP-1 gets released after meals in adults with obesity or type 2 diabetes mellitus compared to adults with normal glucose tolerance. So having less GLP-1 circulating in your blood means you don’t feel as full after eating and get hungry again sooner compared to people who produce more.
GLP-1 has a very short half-life of about two minutes. So GLP-1RA medications were designed to have a very long half-life of about seven days. That’s why semaglutide is given as a weekly injection.
What can users expect? What does the research say?
Higher doses of semaglutide are prescribed to treat obesity compared to type 2 diabetes management (up to 2.4mg versus 2.0mg weekly).
A large group of randomised controlled trials, called STEP trials, all tested weekly 2.4mg semaglutide injections versus different interventions or placebo drugs.
Trials lasting 1.3–2 years consistently found weekly 2.4 mg semaglutide injections led to 6–12% greater weight loss compared to placebo or alternative interventions. The average weight change depended on how long medication treatment lasted and length of follow-up.
Higher doses of semaglutide are prescribed for obesity than for type 2 diabetes. fcm82/Shutterstock Weight reduction due to semaglutide also leads to a reduction in systolic and diastolic blood pressure of about 4.8 mmHg and 2.5 mmHg respectively, a reduction in triglyceride levels (a type of blood fat) and improved physical function.
Another recent trial in adults with pre-existing heart disease and obesity, but without type 2 diabetes, found adults receiving weekly 2.4mg semaglutide injections had a 20% lower risk of specific cardiovascular events, including having a non-fatal heart attack, a stroke or dying from cardiovascular disease, after three years follow-up.
Who is eligible for semaglutide?
Australia’s regulator, the Therapeutic Goods Administration (TGA), has approved semaglutide, sold as Ozempic, for treating type 2 diabetes.
However, due to shortages, the TGA had advised doctors not to start new Ozempic prescriptions for “off-label use” such as obesity treatment and the Pharmaceutical Benefits Scheme doesn’t currently subsidise off-label use.
The TGA has approved Wegovy to treat obesity but it’s not currently available in Australia.
When it’s available, doctors will be able to prescribe semaglutide to treat obesity in conjunction with lifestyle interventions (including diet, physical activity and psychological support) in adults with obesity (a BMI of 30 or above) or those with a BMI of 27 or above who also have weight-related medical complications.
What else do you need to do during Ozempic treatment?
Checking details of the STEP trial intervention components, it’s clear participants invested a lot of time and effort. In addition to taking medication, people had brief lifestyle counselling sessions with dietitians or other health professionals every four weeks as a minimum in most trials.
Support sessions were designed to help people stick with consuming 2,000 kilojoules (500 calories) less daily compared to their energy needs, and performing 150 minutes of moderate-to-vigorous physical activity, like brisk walking, dancing and gardening each week.
STEP trials varied in other components, with follow-up time periods varying from 68 to 104 weeks. The aim of these trials was to show the effect of adding the medication on top of other lifestyle counselling.
Trial participants also exercised for 150 minutes a week. Elena Nichizhenova/Shutterstock A review of obesity medication trials found people reported they needed less cognitive behaviour training to help them stick with the reduced energy intake. This is one aspect where drug treatment may make adherence a little easier. Not feeling as hungry and having environmental food cues “switched off” may mean less support is required for goal-setting, self-monitoring food intake and avoiding things that trigger eating.
But what are the side effects?
Semaglutide’s side-effects include nausea, diarrhoea, vomiting, constipation, indigestion and abdominal pain.
In one study these led to discontinuation of medication in 6% of people, but interestingly also in 3% of people taking placebos.
More severe side-effects included gallbladder disease, acute pancreatitis, hypoglycaemia, acute kidney disease and injection site reactions.
To reduce risk or severity of side-effects, medication doses are increased very slowly over months. Once the full dose and response are achieved, research indicates you need to take it long term.
Given this long-term commitment, and associated high out-of-pocket cost of medication, when it comes to taking semaglutide to treat obesity, there is no way it can be considered “cheating”.
Read the other articles in The Conversation’s Ozempic series here.
Clare Collins, Laureate Professor in Nutrition and Dietetics, University of Newcastle
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails: