7 Days Of Celery Juice: What’s The Verdict?
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Laura “Try” tries many popular trends, and reports on the benefits (or problems, or both). In this case, it’s 7 days of celery juice… Not as a fast, though, i.e. she doesn’t just have celery juice for 7 days, but rather, it’s how she kicks off each morning, with half a liter (16oz) on an empty stomach.
What she found
First, she bought a masticating juicer and organic celery. So, those are expenses to consider, especially the one-off expense of the juicer, and the ongoing expense of organic celery—estimated $90/month).
In terms of taste, she was surprised it wasn’t as bitter as expected, but from the second day onwards, she did use the juicer’s filter to remove the frothy sludge, and she also switched to juicing only the stalks, not the leaves—which are more bitter.
10almonds note: the leaves are more bitter because that’s where the polyphenols are more densely concentrated. The leaves are better for you than the stalks. Enjoy the leaves. Really: if you chop them finely you can use them as herbs in your cooking, and if you’re making a salad, just chop them into that too.
The reason she picked the quantity of half a liter is because this is what she found recommended to coat the stomach lining—on the promise of increased stomach acid production, reduced bacteria overgrowth, as well as antiviral, antifungal, and anti-inflammatory properties. As she’s just one woman without a personal lab, she couldn’t test and thus verify any of these though—but she did still have benefits to report:
She did experience clearer skin, more energy, and better sleep after a few days.
Ultimately, she decided to continue to do it just at the weekends, due to its positive effects, despite the cost and time consumption.
For more personal insights, enjoy:
Click Here If The Embedded Video Doesn’t Load Automatically!
Want to learn more?
You might also like to read:
Enjoy Bitter Foods For Your Heart & Brain
Take care!
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Recommended
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails:
-
From Cucumbers To Kindles
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
You’ve Got Questions? We’ve Got Answers!
Q: Where do I get cucumber extract?
A: You can buy it from BulkSupplements.com (who, despite their name, start at 100g packs)
Alternatively: you want it as a topical ointment (for skin health) rather than as a dietary supplement (for bone and joint health), you can extract it yourself! No, it’s not “just juice cucumbers”, but it’s also not too tricky.
Share This Post
-
Move over, COVID and Flu! We Have “Hybrid Viruses” To Contend With Now
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
Move over, COVID and Flu! We have “hybrid viruses” to contend with now
COVID and influenza viruses can be serious, of course, so let’s be clear up front that we’re not being dismissive of those. But, most people are hearing a lot about them, whereas respiratory syncytial virus (RSV) has flown under a lot of radars.
Simply put, until recently it hasn’t been considered much of a threat except to the young, the old, or people with other respiratory illnesses. Only these days, the prevalence of “other respiratory illnesses” is a lot higher than it used to be!
It’s not just a comorbidity
It’s easy to think “well of course if you have more than one illness at once, especially similar ones, that’s going to suck” but it’s a bit more than that; it produces newer, more interesting, hybrid viruses. Here’s a research paper from last year’s “flu season”:
Coinfection by influenza A virus and respiratory syncytial virus produces hybrid virus particles
Best to be aware of this if you’re in the “older” age-range
It’s not just that the older we are, the more likely we are to get it. Critically, the older we are, the more likely we are to be hospitalized by it.
And..the older we are, the less likely we are to come back from hospital if hospitalized by it.
Some years back, the intensive care and mortality rates for people over the age of 65 were 8% and 7%, respectively:
Respiratory syncytial virus infection in elderly and high-risk adults
…but a new study this year has found the rates like to be 2.2x that, i.e. 15% intensive care rate and 18% mortality, respectively:
Want to know more?
Here are some hot-off-the-press news articles on the topic:
- Better awareness of RSV in older adults is needed to reduce hospitalizations
- Is there also a connection between RSV and asthma?
- Respiratory syncytial virus coinfections conspire to worsen disease
And as for what to do…
Same general advice as for COVID and Flu, just, ever-more important:
- Try to keep to well-ventilated places as much as possible
- Get any worrying symptoms checked out quickly
- Mask up when appropriate
- Get your shots as appropriate
See also:
Harvard Health Review | Fall shots: Who’s most vulnerable to RSV, COVID, and the flu, and which shots are the right choice for you to help protect against serious illness and hospitalization?
Stay safe!
Share This Post
-
How Not To Die – by Dr. Michael Greger
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
We previously reviewed this book some years ago, but we’re revisiting it now because:
- It really is a book that should be in every healthspan-enjoyer’s collection
- Our book reviews back then were not as comprehensive as now (though we still generally try to fit into the “it takes about one minute to read this review” idea, sometimes we’ll spend a little extra time).
Dr. Greger (of “Dr. Greger’s Daily Dozen” fame) outlines for us in cold hard facts and stats what’s most likely to be our cause of death. While this is not a cheery premise for a book, he then sets out to work back from there—what could have prevented those specific things?
Thus, while the book doesn’t confer immortality (the title is not “how to not die”, after all), it does teach us how not to die—i.e, from heart disease, lung diseases, brain diseases, digestive cancers, infections, diabetes, high blood pressure, liver disease, blood cancers, kidney disease, breast cancer, suicidal depression, prostate cancer, Parkinson’s disease, and even iatrogenic causes.
This it does with a lot of solid science, explained for the layperson, and/but without holding back when it comes to big words, and a lot of them, at that. If you want to add in daily exercises, just lifting the book could be a start; weighing in at 678 pages, it’s an information-dense tome that’s more likely to be sifted through than read cover-to-cover.
The style is thus dense science somewhat editorialized for lay readability, and well-evidenced with around 3,000 citations. That’s not a typo; there are 178 pages of bibliography at the back with about 15–20 scientific references per page.
In terms of practical use, he does also devote chapters to that, it’s not just all textbook. Indeed, he discusses the reasonings behind the items, portion sizes, and quantities of his “daily dozen” foods, so that the reader will understand how much bang-for-buck they deliver, and then it’ll seem a lot less like an arbitrary list, and more likely to be adopted and maintained.
Bottom line: if you care about not getting life-threatening illnesses (which at the end of the day, come to most people at some point), then this book is a must-read.
Share This Post
Related Posts
-
52 Weeks to Better Mental Health – by Dr. Tina Tessina
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
We’ve written before about the health benefits of journaling, but how to get started, and how to make it a habit, and what even to write about?
Dr. Tessina presents a year’s worth of journaling prompts with explanations and exercises, and no, they’re not your standard CBT flowchart things, either. Rather, they not only prompt genuine introspection, but also are crafted to be consistently uplifting—yes, even if you are usually the most disinclined to such positivity, and approach such exercises with cynicism.
There’s an element of guidance beyond that, too, and as such, this book is as much a therapist-in-a-book as you might find. Of course, no book can ever replace a competent and compatible therapist, but then, competent and compatible therapists are often harder to find and can’t usually be ordered for a few dollars with next-day shipping.
Bottom line: if undertaken with seriousness, this book will be an excellent investment in your mental health and general wellbeing.
Click here to check out 52 Weeks to Better Mental Health, and get on the best path for you!
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails:
-
Ozempic’s cousin drug liraglutide is about to get cheaper. But how does it stack up?
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
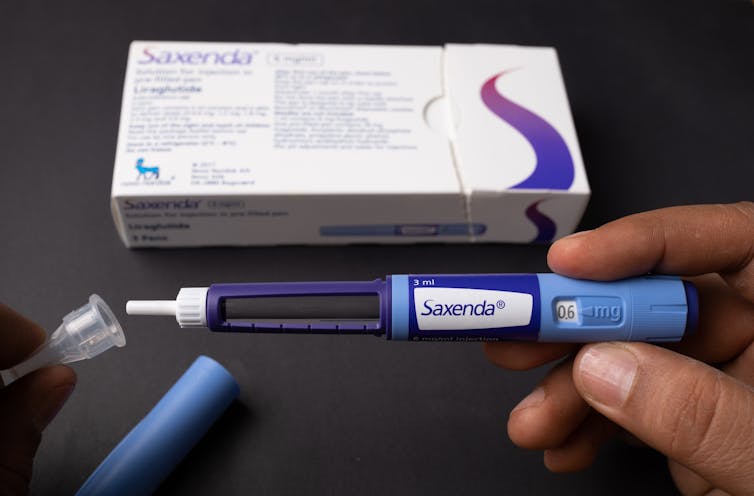
Fourteen years ago, the older drug cousin of semaglutide (Ozempic and Wegovy) came onto the market. The drug, liraglutide, is sold under the brand names Victoza and Saxenda.
Patents for Victoza and Saxenda have now expried. So other drug companies are working to develop “generic” versions. These are likely be a fraction of current cost, which is around A$400 a month.
So how does liraglutide compare with semaglutide?
Halfpoint/Shutterstock How do these drugs work?
Liraglutide was not originally developed as a weight-loss treatment. Like semaglutide (Ozempic), it originally treated type 2 diabetes.
The class of drugs liraglutide and semaglutide belong to are known as GLP-1 mimetics, meaning they mimic the natural hormone GLP-1. This hormone is released from your small intestines in response to food and acts in several ways to improve the way your body handles glucose (sugar).
How do they stop hunger?
Liraglutide acts in several regions of the unconscious part of your brain, specifically the hypothalamus, which controls metabolism, and parts of the brain stem responsible for communicating your body’s nutrient status to the hypothalamus.
Its actions here appear to reduce hunger in two different ways. First, it helps you to feel full earlier, making smaller meals more satisfying. Second, it alters your “motivational salience” towards food, meaning it reduces the amount of food you seek out.
Liraglutide’s original formulation, designed to treat type 2 diabetes, was marketed as Victoza. Its ability to cause weight loss was evident soon after it entered the market.
Shortly after, a stronger formulation, called Saxenda, was released, which was intended for weight loss in people with obesity.
How much weight can you lose with liraglutide?
People respond differently and will lose different amounts of weight. But here, we’ll note the average weight loss users can expect. Some will lose more (sometimes much more), others will lose less, and a small proportion won’t respond.
The first GLP-1 mimicking drug was exenatide (Bayetta). It’s still available for treating type 2 diabetes, but there are currently no generics. Exenatide does provide some weight loss, but this is quite modest, typically around 3-5% of body weight.
For liraglutide, those using the drug to treat obesity will use the stronger one (Saxenda), which typically gives about 10% weight loss.
Semaglutide, with the stronger formulation called Wegovy, typically results in 15% weight loss.
The newest GLP-1 mimicking drug on the market, tirzepatide (Mounjaro for type 2 diabetes and Zepbound for weight loss), results in weight loss of around 25% of body weight.
What happens when you stop taking them?
Despite the effectiveness of these medications in helping with weight loss, they do not appear to change people’s weight set-point.
So in many cases, when people stop taking them, they experience a rebound toward their original weight.
People often regain weight when when they stop taking the drug. Mohammed_Al_Ali/Shutterstock What is the dose and how often do you need to take it?
Liraglutide (Victoza) for type 2 diabetes is exactly the same drug as Saxenda for weight loss, but Saxenda is a higher dose.
Although the target for each formulation is the same (the GLP-1 receptor), for glucose control in type 2 diabetes, liraglutide has to (mainly) reach the pancreas.
But to achieve weight loss, it has to reach parts of the brain. This means crossing the blood-brain barrier – and not all of it makes it, meaning more has to be taken.
All the current formulations of GLP-1 mimicking drug are injectables. This won’t change when liraglutide generics hit the market.
However, they differ in how frequently they need to be injected. Liraglutide is a once-daily injection, whereas semaglutide and tirzepatide are once-weekly. (That makes semaglutide and tirzepatide much more attractive, but we won’t see semaglutide as a generic until 2033.)
What are the side effects?
Because all these medicines have the same target in the body, they mostly have the same side effects.
The most common are a range of gastrointestinal upsets including nausea, vomiting, bloating, constipation and diarrhoea. These occur, in part, because these medications slow the movement of food out of the stomach, but are generally managed by increasing the dose slowly.
Recent clinical data suggests the slowing in emptying of the stomach can be problematic for some people, and may increase the risk of of food entering the lungs during operations, so it is important to let your doctor know if you are taking any of these drugs.
Because these are injectables, they can also lead to injection-site reactions.
Gastrointestinal side effects are most common. Halfpoint/Shutterstock During clinical trials, there were some reports of thyroid disease and pancreatitis (inflammation of the pancreas). However, it is not clear that these can be attributed to GLP-1 mimicking drugs.
In animals, GLP-1 mimicking drugs drugs have been found to negatively alter the growth of the embryo. There is currently no controlled clinical trial data on their use during pregnancy, but based on animal data, these medicines should not be used during pregnancy.
Who can use them?
The GLP-1 mimicking drugs for weight loss (Wegovy, Saxenda, Zepbound/Mounjaro) are approved for use by people with obesity and are meant to only be used in conjunction with diet and exercise.
These drugs must be prescribed by a doctor and for obesity are not covered by the Pharmaceutical Benefits Scheme, which is one of the reasons why they are expensive. But in time, generic versions of liraglutide are likely to be more affordable.
Sebastian Furness, ARC Future Fellow, School of Biomedical Sciences, The University of Queensland
This article is republished from The Conversation under a Creative Commons license. Read the original article.
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails:
-
Reporting on psychedelics research or legislation? Proceed with caution
10almonds is reader-supported. We may, at no cost to you, receive a portion of sales if you purchase a product through a link in this article.
More cities and states are introducing bills to decriminalize and regulate access to psychedelic drugs, which could potentially become another option to treat mental health conditions and substance use disorders. But the substances remain illegal under U.S. federal law and scientific evidence about their effectiveness is still far from conclusive.
This month alone, California lawmakers introduced a bill to allow people 21 and older to consume psychedelic mushrooms under medical supervision. In Massachusetts, lawmakers are working on a bill that would legalize psilocybin, the active ingredient of psychedelic mushrooms. And Arizona legislators have also introduced a bill that would make psychedelic mushrooms available as a mental health treatment option.
Last December, Congress passed legislation that included funding for psychedelic clinical trials for active-duty service members. And in January this year, the Department of Veterans Affairs announced that it will begin funding research on MDMA, also known as ecstasy, and psilocybin, to treat veterans with post-traumatic stress disorder and depression. This is the first time since the 1960s that the VA is funding research on such compounds, according to the department.
The rise of proposed and passed legislation in recent years necessitates more journalistic coverage. But it’s important for journalists to go beyond what the bills and lawmakers say and include research studies about psychedelics and note the limitations of those studies.
Major medical organizations, including the American Psychiatric Association, have not yet endorsed psychedelics to treat psychiatric disorders, except in clinical trials, due to inadequate scientific evidence.
The authors of a 2023 study published in the journal Therapeutic Advances in Psychopharmacology, also advise “strong caution” regarding the hype around the potential medical use of psychedelics. “There is not enough robust evidence to draw any firm conclusions about the safety and efficacy of psychedelic therapy,” they write.
Scientists are still trying to better understand how psychedelics work, what’s the best dose for treating different mental health conditions and how to reduce the risk of potential side effects such as intense emotional experiences or increased heart rate and blood pressure, the authors of a February 2024 study published in the journal Progress in Neuro-Psychopharmacology and Biological Psychiatry write.
In a 2022 study published in JAMA Psychiatry, Dr. Joshua Siegel and his colleagues at Washington University in St. Louis write that while legislative reform for psychedelic drugs is moving forward rapidly, several issues have not been addressed, including:
- A mechanism for verifying the chemical content of drugs that are obtained from outside the medical establishment.
- Licensure and training criteria for practitioners who wish to provide psychedelic treatment.
- Clinical and billing infrastructure.
- Assessing potential interactions with other drugs.
- How the drugs should be used in populations such as youths, older adults and pregnant people.
“Despite the relative rapidity with which some have embraced psychedelics as legitimate medical treatments, critical questions about the mechanism of action, dose and dose frequency, durability of response to repeated treatments, drug-drug interactions, and the role that psychotherapy plays in therapeutic efficacy remain unanswered,” Siegel and colleagues write.
What are psychedelics?
Psychedelics are among the oldest class of mind-altering substances, used by humans for thousands of years in traditional or religious rituals.
In 2021, 74 million people 12 years and older reported using hallucinogens, according to the National Survey on Drug Use and Health.
The terms “psychedelics” and “hallucinogens” are used interchangeably in public discourse, but scientifically, hallucinogens fall into three groups based on chemical structure and mechanism of action, according to NIH’s National Institute on Drug Abuse:
- Psychedelic drugs, also called “classic psychedelics” or simply “psychedelics,” mainly affect the way the brain processes serotonin, a chemical that carries messages between nerve cells in the brain and the body. These drugs can bring on vivid visions and affect a person’s sense of self, according to NIDA. Drugs in this category include:
- Psilocybin is the active ingredient in psychedelic mushrooms, also known as “magic” mushrooms or shrooms. It’s a Schedule 1 drug in the U.S. under the Controlled Substances Act, which means it has a high potential for abuse and has no accepted medical use. However, some states have decriminalized it, according to NIDA. The drug has also been given the Breakthrough Therapy designation from the FDA, a process to speed up the development and review of drugs, for the treatment of major depressive disorder.
- LSD, or lysergic acid diethylamide, is a synthetic chemical made from a fungus that infects rye. It’s a Schedule 1 drug.
- DMT, or dimethyltryptamine, found in certain plants native to the Amazon rainforest, has been used in religious practices and rituals. The plants are sometimes used to make a tea called ayahuasca. DMT can also be made in the lab as a white powder. DMT is generally smoked or consumed in brews like ayahuasca. It’s a Schedule 1 drug.
- Mescaline, a chemical compound found in a small cactus called peyote, has been used by Indigenous people in northern Mexico and the southwestern U.S. in religious rituals. Mescaline can also be produced in the lab. Mescaline and peyote are Schedule 1 drugs.
- Dissociative drugs affect how the brain processes glutamate, an abundant chemical released by nerve cells in the brain that plays an important role in learning and memory. These drugs can make people feel disconnected from their bodies and surroundings. Drugs in this category include:
- PCP, or phencyclidine, was developed in the 1950s as an injectable anesthetic but was discontinued because patients became agitated and delusional. Today it is an illegal street drug. It’s a Schedule 2 drug, which means it has a high potential for abuse, but lower compared to Schedule 1 drugs.
- Ketamine, a drug developed in the 1960s and used as an anesthetic in the Vietnam War, is approved by the FDA as an anesthetic. It has been shown to play a role in pain management and treatment of depression. It is also illegally used for its hallucinogenic effects. It is a Schedule 3 drug, which means it has a moderate to low potential for physical and psychological dependence. A chemically-similar drug called esketamine is approved by the FDA for the treatment of depression that doesn’t respond to standard treatment.
- Other hallucinogens, which affect different brain functions and can cause psychedelic and potentially dissociative effects, include:
- MDMA, or ecstasy, is a synthetic drug that’s a stimulant and hallucinogen. It is a Schedule 1 drug. It has been given the Breakthrough Therapy designation from the FDA for the treatment of PTSD.
- Salvia is an herb in the mint family that has hallucinogenic effects. It is not a federally controlled drug, but it is controlled in some states, according to the DEA.
- Ibogaine is derived from the root bark of a West African shrub and is a stimulant and hallucinogen. It is a Schedule 1 drug.
Research on psychedelics
There was a wave of studies on psychedelics, particularly LSD, in the 1950s and 1960s, but they came to a halt when the U.S. declared a “War on Drugs” in 1971 and tightened pharmaceutical regulations. There was little research activity until the early 1990s when studies on drugs such as MDMA and DMT began to emerge.
In 2006, researchers at Johns Hopkins University published a seminal double-blind study in which two-thirds of participants — who had never taken psychedelics previously — said their psychedelic sessions were among the most meaningful experiences of their lives.
“These studies, among others, renewed scientific interest in psychedelics and, accordingly, research into their effects has continued to grow since,” Jacob S. Aday and colleagues write in a 2019 study published in Drug Science, Policy and Law.
In their paper, Aday and colleagues argue that 2018 may be remembered as the true turning point in psychedelic research due to “advances within science, increased public interest, and regulatory changes,” such as psilocybin receiving the “breakthrough therapy” status from the FDA.
Today, there are numerous ongoing clinical trials on the therapeutic potential of psychedelics for different conditions, including substance use disorders and mental health conditions such as depression, anxiety and post-traumatic stress disorder.
Given the growing number of studies on psychedelics, the Food and Drug Administration issued a draft guidance in June 2023 for clinical trials with psychedelic drugs, aiming to help researchers design studies that will yield more reliable results for drug development.
The systematic reviews highlighted below show that there’s a lack of robust study designs in many psychedelic clinical trials. Some have small sample sizes. Some include participants who have used psychedelics before, so when they participate in a randomized controlled clinical trial, they know whether they are receiving psychedelic treatment or a placebo. Or, some include participants who may have certain expectations due to positive coverage in the lay media, hence creating bias in the results.
If you’re covering a study about psychedelics…
It’s important for journalists to pay close attention to study design and speak with an expert who is not involved in the study.
In a February 2024 blog post from Harvard Law School’s Petrie-Flom Center, Leiden University professors Eiko I. Fried and Michiel van Elk share several challenges in psychedelic research:
- “Conclusions are dramatically overstated in many studies. This ranges from conclusions in the results sections, abstracts, and even titles of papers not consistent with the reported results.”
- “There is emerging evidence that adverse events resulting from psychedelic substances are both common and underreported.”
- Some studies don’t have control groups, which can create problems for interpreting results, “because treatments like psychedelics need to be compared against a placebo or other treatment to conclude that they work beyond the placebo effect or already existing, readily available treatments.”
- “Participants in psychedelic studies usually know if they are in the treatment or control group, which artificially increases the apparent efficacies of psychedelics in clinical studies.”
- Small sample sizes can affect the statistical power and generalizability of the findings. “Small samples also mean that results are not representative. For example, participants with severe or comorbid mental health problems are commonly excluded from psychedelic studies, and therefore results may look better in these studies than in real-world psychiatric settings.”
- Many studies do not include long-term follow-ups of participants. “Studying how these people are feeling a few days or weeks after they receive treatment is not sufficient to establish that they are indeed cured from depression.”
Fried and van Elk also have a useful checklist for assessing the quality and scientific rigor of psychedelic research in their 2023 study “History Repeating: Guidelines to Address Common Problems in Psychedelic Science,” published in the journal Therapeutic Advances in Psychopharmacology.
Journalists should also remind their audiences that the drugs are still illegal under federal law and can pose a danger to health.
In California, the number of emergency room visits involving the use of hallucinogens increased by 54% between 2016 and 2022, according to a January 2024 study published in Addiction. Meanwhile, the law enforcement seizure of psychedelic mushrooms has risen dramatically, increasing nearly four-fold between 2017 and 2022, according to a February 2024 study published in the journal Drug and Alcohol Dependence.
Below, we have curated and summarized five recent studies, mostly systematic reviews and meta-analyses, which examine various aspects of psychedelic drugs, including legislative reform; long-term effects; efficacy and safety for the treatment of anxiety, depression and PTSD; and participation of older adults in clinical trials. The research summaries are followed by recommended reading.
Research roundup
Psychedelic Drug Legislative Reform and Legalization in the US
Joshua S. Siegel, James E. Daily, Demetrius A. Perry and Ginger E. Nicol. JAMA Psychiatry, December 2022.The study: Most psychedelics are Schedule I drugs federally, but state legislative reforms are changing the prospects of the drugs’ availability for treatment and their illegal status. For a better understanding of the legislative reform landscape around Schedule I psychedelic drugs, researchers collected all bills and ballot initiatives related to psychedelic drugs that were introduced into state legislatures between 2019 and September 2022. They used publicly available sources, including BillTrack50, Ballotpedia and LexisNexis.
The findings: In total, 25 states considered 74 bills, although the bills varied widely in their framework. A majority proposed decriminalization but only a few would require medical oversight and some would not even require training or licensure, the authors write. Ten of those bills became law in seven states — Colorado, Connecticut, Hawaii, New Jersey, Oregon, Texas and Washington. As of August 1, 2022, 32 bills were dead and 32 remained active.
The majority of the bills — 67 of them — referred to psilocybin; 27 included both psilocybin and MDMA; 43 proposed decriminalization of psychedelic drugs.
To predict the future legalization of psychedelics, the authors also created two models based on existing medical and recreational marijuana reform. Using 2020 as the year of the first psychedelic decriminalization in Oregon, their models predict that 26 states will legalize psychedelics between 2033 and 2037.
In the authors’ words: “Despite the relative rapidity with which some have embraced psychedelics as legitimate medical treatments, critical questions about the mechanism of action, dosing and dose frequency, durability of response to repeated treatments, drug-drug interactions, and the role psychotherapy plays in therapeutic efficacy remain unanswered. This last point is critical, as a significant safety concern associated with drugs like psilocybin, MDMA, or LSD is the suggestibility and vulnerability of the patient while under the influence of the drug. Thus, training and clinical oversight is necessary to ensure safety and also therapeutic efficacy for this divergent class of treatments.”
Who Are You After Psychedelics? A Systematic Review and a Meta-Analysis of the Magnitude of Long-Term Effects of Serotonergic Psychedelics on Cognition/Creativity, Emotional Processing and Personality
Ivana Solaja, et al. Neuroscience & Behavioral Reviews, March 2024.The study: Many anecdotal reports and observational studies have reported that psychedelics, even at microdoses, which are roughly one-tenth of a typical recreational dose, may enhance certain aspects of cognition and/or creativity, including coming up with new, useful ideas. Cognition is a “range of intellectual functions and processes involved in our ability to perceive, process, comprehend, store and react to information,” the authors explain. There are established relationships between impaired cognitive functioning and mental health disorders.
Due to limitations such as a lack of rigorous study designs, various populations in the studies and lack of documented dosage, it’s difficult to draw any conclusions about changes that last at least one week as a result of consuming psychedelics.
The authors screened 821 studies and based on the criteria they had set, found 10 to be eligible for the review and meta-analysis. The drugs in the studies include psilocybin, ayahuasca and LSD.
The findings: Overall, there was little evidence that these psychedelics have lasting effects on creativity. Also, there was not sufficient evidence to determine if this group of psychedelics enhances cognition and creativity in healthy populations or improves cognitive deficits in the study populations.
Pooled data from three studies showed lasting improvement in emotional processing — perceiving, expressing and managing emotions.
The studies offered little evidence suggesting lasting effects of psychedelics on personality traits.
In the authors’ words: “Results from this study showed very limited evidence for any lasting beneficial effects across these three psychological constructs. However, preliminary meta-analytic evidence suggested that these drugs may have the potential to cause lasting improvement in emotional recognition time. Future studies investigating these constructs should employ larger sample sizes, better control conditions, standardized and validated measures and longer-term follow-ups.”
The Impact of Psychedelics on Patients with Alcohol Use Disorder: A Systematic Review with Meta-Analysis
Dakota Sicignano, et al. Current Medical Research and Opinion, December 2023.The study: Researchers are exploring the psychedelics’ potential for the treatment of alcohol use disorder, which affected nearly 30 million Americans in 2022. The authors of this study searched PubMed from 1960 to September 2023 for studies on the use of psychedelics to treat alcohol use disorder. Out of 174 English-language studies, they selected six studies that met the criteria for their analysis.
The findings: LSD and psilocybin are promising therapies for alcohol use disorder, the authors report. However, five of the six trials were conducted in the 1960s and 1970s and may not reflect the current treatment views. Also, four of the six studies included patients who had used psychedelics before participating in the study, increasing the risk of bias.
In the authors’ words: “Despite the existence of several clinical trials showing relatively consistent benefits of psychedelic therapy in treating alcohol use disorder, there are important limitations in the dataset that must be appreciated and that preclude a conclusive determination of its value for patient care at this time.”
Older Adults in Psychedelic-Assisted Therapy Trials: A Systematic Review
Lisa Bouchet, et al. Journal of Psychopharmacology, January 2024.The study: People 65 years and older have been underrepresented in clinical trials involving psychedelics, including the use of psilocybin for the treatment of depression and anxiety. About 15% of adults older than 60 suffer from mental health issues, the authors note. They wanted to quantify the prevalence of older adults enrolled in psychedelic clinical trials and explore safety data in this population. They searched for English-language studies in peer-reviewed journals from January 1950 to September 2023. Of 4,376 studies, the authors selected 36. The studies involved psilocybin, MDMA, LSD, ayahuasca, and DPT (dipropyltryptamine), which is a less-studied synthetic hallucinogen.
The findings: Of the 1,400 patients participating in the selected studies, only 19 were 65 and older. Eighteen received psychedelics for distress related to cancer or other life-threatening illnesses. In a trial of MDMA-assisted therapy for PTSD, only one older adult was included. Adverse reactions to the drugs among older patients, including heart and gastrointestinal issues were resolved within two days and didn’t have a long-lasting impact.
In the authors’ words: “Although existing data in older adults is limited, it does provide preliminary evidence for the safety and tolerability of [psychedelic-assisted therapy] in older patients, and as such, should be more rigorously studied in future clinical trials.”
Efficacy and Safety of Four Psychedelic-Assisted Therapies for Adults with Symptoms of Depression, Anxiety, and Posttraumatic Stress Disorder: A Systematic Review and Meta-Analysis
Anees Bahji, Isis Lunsky, Gilmar Gutierrez and Gustavo Vazquez. Journal of Psychoactive Drugs, November 2023.The study: LSD, psilocybin, ayahuasca and MDMA have been approved for clinical trials on psychedelic-assisted therapy of mental health conditions in Canada and the U.S. However, major medical associations, including the American Psychiatric Association, have argued that there is insufficient scientific evidence to endorse these drugs for treating mental health disorders. To better understand the current evidence, researchers reviewed 18 blinded, randomized controlled trials, spanning 2008 through 2023. Most studies were conducted in the U.S. or Switzerland.
The findings: The studies overall suggest preliminary evidence that psychedelic drugs are mostly well-tolerated. Psilocybin and MDMA therapies may offer relief from depression and PTSD symptoms for at least a year. Most studies also used therapy and psychological support along with psychedelics.
In the authors’ words: “Despite the promising evidence presented by our study and previous reviews in the field, the evidence base remains limited and underpowered. Long-term efficacy and safety data are lacking,” the authors write. “Future steps should encourage and highlight the need for more robust larger scale randomized controlled trials with longer follow-up periods, and efforts to address regulatory and legal barriers through the collaborations between researchers, healthcare professionals, regulatory bodies, and policymakers.”
This article first appeared on The Journalist’s Resource and is republished here under a Creative Commons license.
Don’t Forget…
Did you arrive here from our newsletter? Don’t forget to return to the email to continue learning!
Learn to Age Gracefully
Join the 98k+ American women taking control of their health & aging with our 100% free (and fun!) daily emails: